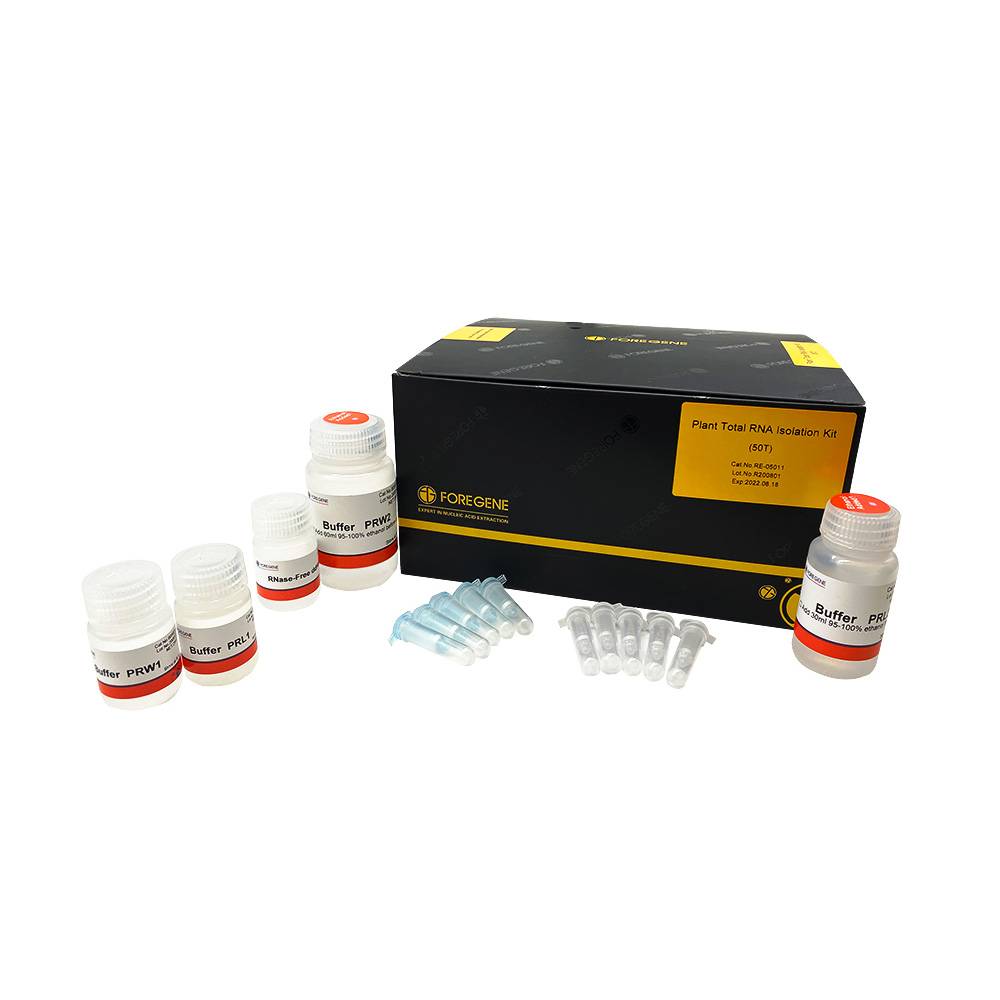
Plant Total RNA Isolation Kit Total RNA Purificaiton Kit for Plant Low in Polysaccharides and Polyphenols
Specifications
50 Preps, 200 Preps
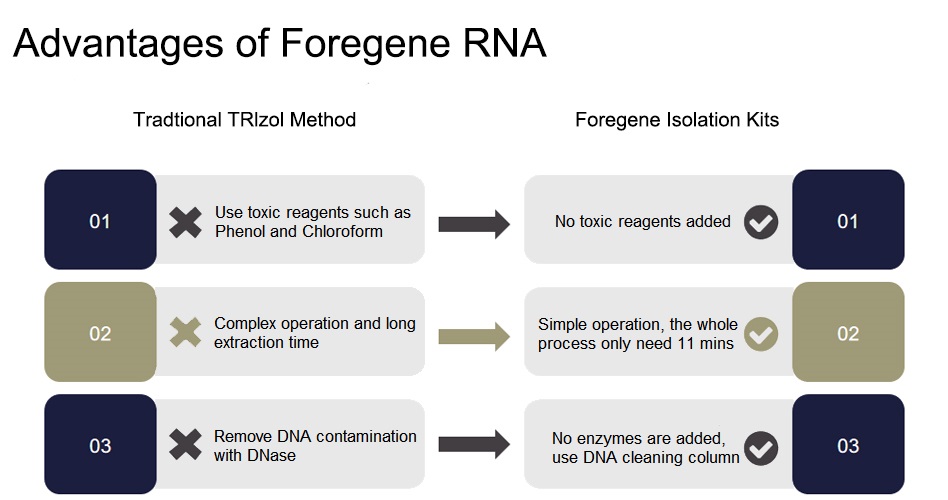
The kit uses the spin column and formula developed by Foregene, which can efficiently extract high-purity and high-quality total RNA from various plant tissues with low polysaccharides and polyphenols content. For plant samples with high polysaccharides or polyphenols content, it is recommended to use Plant Total RNA Isolation Plus Kit to get better RNA extraction results. The kit provides the DNA-Cleaning column that can easily remove genomic DNA from the supernatant and tissue lysate. RNA-only column can effectively bind RNA. The kit can process a large number of samples at the same time.
The entire system does not contain RNase, so the purified RNA will not be degraded. Buffer PRW1 and Buffer PRW2 can ensure that the RNA obtained is not contaminated by protein, DNA, ions, and organic compounds.
Kit components
|
Buffer PSL1, Buffer PS, Buffer PSL2 |
|
Buffer PRW1, Buffer PRW2 |
|
RNase-Free ddH2O, DNA-Cleaning Column |
|
RNA-Only Column |
|
Instructions |
Features&advantages
■ Operation at room temperature (15-25℃) throughout the whole process, without ice bath and low temperature centrifugation.
■ Complete kit RNase-Free, no need to worry about RNA degradation.
■ DNA-Cleaning Column specifically binds to DNA, so that the kit can remove genomic DNA contamination without adding DNase.
■ High RNA yield: RNA-only Column and unique formula can efficiently purify RNA.
■ Fast speed: easy to operate and can be completed within 30 minutes.
■ Safety: no organic reagent is required.
■ High quality: The purified RNA fragments are of high purity, free of protein and other impurities, and can meet various downstream experimental applications.
Kit application
It is suitable for the extraction and purification of total RNA from fresh or frozen plant tissue samples (especially fresh plant leaf tissue) with low polysaccharide and polyphenol content.
Work flow
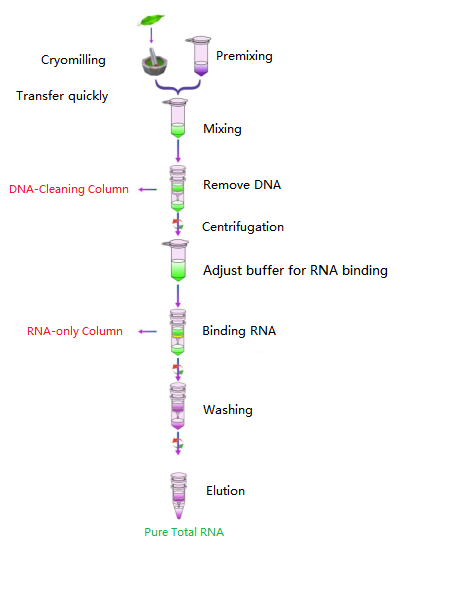
Diagram
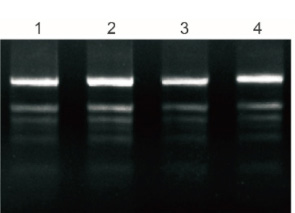
Plant Total RNA Isolation Kit Plus processed 50mg of fresh leaves of polysaccharides and polyphenols, and 5% purified RNA was tested by electrophoresis.
1: Banana
2: Ginkgo
3: Cotton
4: Pomegranate
Storage and shelf life
The kit can be stored for 12 months at room temperature (15–25 ℃) in dry environment, and 2–8 ℃ for longer time(24 months).
Buffer PSL1 can be stored at 4 ℃ for 1 month after adding 2-hydroxy-1-ethanethiol(optional).
Problem Analysis Guide
The following analysis of the problems you may encounter in Plant Total RNA extraction will help you with your experiments. In addition, for other experimental or technical problems in addition to operating instructions and problem analysis, we have dedicated technical support to help you. If you have any needs, please contact us at: 028-83360257 or E-mali : Tech@foregene.com.
The spin column is clogged
Blockage of the spin column will cause the RNA yield to be reduced or even unable to be purified to obtain RNA, and the quality of the obtained RNA will be low.
Common Cause Analysis:
1. The sample is not broken completely.
Incomplete sample fragmentation can block the DNA-Cleaning Column, which can also affect RNA yield and quality. We recommend that when performing sample fragmentation, quickly grind in sufficient amounts of liquid nitrogen to break up the tissues such as cell walls and cell membranes of the samples as much as possible. For plant samples of polyphenol polysaccharides, we recommend that you use Plant Total RNA Isolation Kit Plus.
2.Aspirate the DNA-Cleaning Column isolated supernatant, aspirate the possible cell debris pellet.
Aspirated cell debris pellet can clog the RNA-only Column during RNA adsorption procedures (see step 5 of procedure, step 6 of the polysaccharide polyphenol procedure). We recommend that care must be taken when aspirating this supernatant to avoid cell debris being aspirated.
3. The initial amount of sample is too large.
Excessive sample usage will result in incomplete sample fragmentation or incomplete lysis of cells by Buffer PRL1 or Buffer PSL1, resulting in a clogged purification column for purification operations. Plant Total RNA Isolation Kit has an initial maximum of 50 mg per single purification of an operated sample. For plant samples of polyphenol polysaccharides, we recommend that you try the Plant Total RNA Isolation Kit Plus.
4. The temperature of the centrifuge is too low.
Whole RNA isolation and purification except for liquid nitrogen disruption of sample tissue, all steps are performed at room temperature (20-25 °C). Some low-temperature centrifuges have temperatures below 20 °C, which can cause blockages in the DNA-Cleaning Column and/or RNA-only Column. If this occurs, set the centrifuge temperature to 20-25 °C and pre-warm the lysis mixture and/or the addition of ethanol separation supernatant to 37 °C.
No RNA extracted or RNA yield is low
There are usually many factors that affect the recovery efficiency, such as: sample RNA content, operation method, the elution volume, etc.
Analysis of common causes as below:
1.An ice bath or low temperature (4°C) centrifugation was performed during the operation.
Suggestion: Operate at room temperature (15-25°C) in the whole process, do not do ice bath and low temperature centrifugation.
2.The RNA has been degraded due to improper preservation of the sample or long-term preservation of the sample.
Recommendation: Freshly collected samples should be quickly frozen in liquid nitrogen, and then stored at -80°C for a long time,avoid repeated freezing and thawing of samples; or immediately soak the samples in RNA stabilizer RNAlater solution (animal samples).
3.Insufficient sample fragmentation and lysis lead to blockage of the purification column.
Suggestion: When grinding the tissue, please ensure that the tissue is sufficiently ground, and quickly transfer it to the pre-prepared Buffer PSL1 (confirm that the correct proportion of β-ME has been added, see step 1 of the procedure).
4.The eluent was added incorrectly.
Suggestion: Make sure that RNase-Free ddH2O is dripped into the middle of the purification column membrane.
5.The correct volume of absolute ethanol was not added to Buffer PSL2 or Buffer PRW2.
Suggestion: Please follow the instructions, add the correct volume of absolute ethanol to Buffer PSL2 and Buffer PRW2 and mix well before the kit is used.
6.The amount of tissue sample is inappropriate.
Suggestion: Use 50 mg of tissue per 500 μl of Buffer PSL1. Using too much tissue will reduce the amount of RNA extracted and the purity of the resulting RNA will also be reduced. We strongly recommend that the initial sample dosage should not exceed 50 mg per RNA extraction operation.
7. Inappropriate elution volume or incomplete elution.
Suggestion: The eluent volume of the purification column is 50-200 μl; if the elution effect is not satisfactory, it is recommended to extend the time at room temperature after adding preheated RNase-Free ddH2O, such as 5-10min.
8.The purification column has ethanol residue after washing with Buffer PRW2.
Suggestion: If the empty tube is centrifuged for 1 min and there is still ethanol remaining after washing in Buffer PRW2, you can increase the time of the empty tube centrifugation to 2 min, or place the purification column at room temperature for 5 min to fully remove the residual ethanol.
9.The kit was used incorrectly.
Suggestion: For plant samples of polyphenolic polysaccharides, using common kits such as Plant Total RNA Isolation Kit may not be able to obtain ideal RNA samples. We recommend you to use Plant Total RNA IsolationKit Plus, which is specially designed for polyphenolic polysaccharide plant samples. A kit specially developed for extracting RNA from polyphenol and polysaccharide plant samples.
OD260/OD280 value is low
RNA elution with ddH2O and used for spectrophotometer readings results in low OD260/OD280 values. We recommend using 10 mM Tris-HCl, pH 7.5 (rather than RNase-Free ddH2O to elute RNA) to obtain relatively correct OD260/OD280 values, see “RNA Concentration and Purification Assays” on page 19.
The purified RNA is degraded
The quality of purified RNA is related to factors such as sample preservation, RNase contamination, and manipulation.
Analysis of common causes:
1.Tissue samples were not stored in time after collection.
Recommendation: If the tissue samples are not used in time after collection, please store them in liquid nitrogen at low temperature immediately or transfer them to -80°C for long-term storage after quick freezing in liquid nitrogen, or immediately immerse the samples in RNA stabilizer RNAlater solution (animal samples ). For RNA extraction, try to use freshly collected tissue samples.
2.Repeated freezing and thawing of tissue samples.
Suggestion: When storing tissue samples, it is best to cut them into small pieces for preservation, and take out a part of them when using them to avoid the degradation of RNA caused by repeated freezing and thawing of the samples.
3.RNase is introduced in the operation room or not worn disposable gloves, masks, etc.
Suggestion: RNA extraction experiments are best performed in separate RNA operations, and the laboratory table should be cleaned before the experiment, and disposable gloves and masks should be worn during the experiment to avoid RNA degradation caused by the introduction of RNase to the greatest extent.
4.The reagent is contaminated by RNase during use.
Suggestion: Replace with a new series of plant total RNA extraction kits for related experiments.
5.The centrifuge tubes and pipette tips used for RNA manipulation are contaminated with RNase.
Suggestion: Make sure that the centrifuge tubes, pipette tips, pipettes, etc. used in RNA extraction are all RNase-Free.
Instruction Manuals: